|
Because scandium, yttrium, and lanthanum actually do not form compounds analogous to those of the other transition metals and because their chemistry is quite homologous to that of the lanthanoids, they are excluded from the present discussion of the main transition metals. These three main transition series are included in the set of 30 elements often called the d-block transition metals. The third series extends from lanthanum (symbol La, atomic number 57) to mercury (symbol Hg, atomic number 80). The second series includes the elements yttrium (symbol Y, atomic number 39) to cadmium (symbol Cd, atomic number 48). The first main transition series begins with either scandium (symbol Sc, atomic number 21) or titanium (symbol Ti, atomic number 22) and ends with zinc (symbol Zn, atomic number 30). The transition metals may be subdivided according to the electronic structures of their atoms into three main transition series, called the first, second, and third transition series, and two inner transition series, called the lanthanoids and the actinoids. 
Without exception, the elements of the main transition series (i.e., excluding the lanthanoids and actinoids as specified below) form stable compounds in two or more formal oxidation states.įacts You Should Know: The Periodic Table Quiz Third, most of these elements dissolve in mineral acids, although a few, such as platinum, silver, and gold, are called “noble”-that is, are unaffected by simple (nonoxidizing) acids. Second, the transition metals form many useful alloys, with one another and with other metallic elements. Many of the elements are technologically important: titanium, iron, nickel, and copper, for example, are used structurally and in electrical technology. 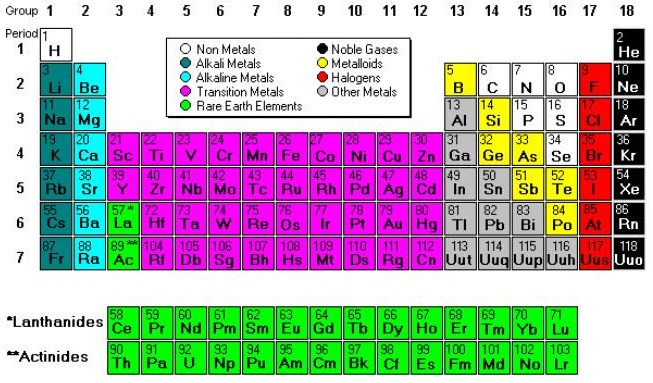
The range in these properties is considerable therefore the statements are comparative with the general properties of all the other elements. The most striking similarities shared by the 24 elements in question are that they are all metals and that most of them are hard, strong, and lustrous, have high melting and boiling points, and are good conductors of heat and electricity. Specifically, they form Groups 3 (IIIb) through 12 (IIb). They occupy the middle portions of the long periods of the periodic table of elements between the groups on the left-hand side and the groups on the right. While the term transition has no particular chemical significance, it is a convenient name by which to distinguish the similarity of the atomic structures and resulting properties of the elements so designated. Transition metal, any of various chemical elements that have valence electrons-i.e., electrons that can participate in the formation of chemical bonds-in two shells instead of only one. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians. 
COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
